So What Are Alkaloids Exactly?
Alkaloids are a class of naturally occurring organic compounds that contain at least one nitrogen atom (although it should be noted that the line between alkaloids and other compounds which contain nitrogen is not clear-cut). In essence, alkaloids are organic alkaline compounds. Alkaline is a chemistry term and is another word for basic (as opposed to acidic).
Alkaloids are produced by many different organisms like bacteria, fungi, plants, and animals. When it comes to plants, over 10,000 alkaloids have been isolated from over 300 plant families. Humans have used alkaloids for thousands of years for medicinal, recreational, and even ritual purposes. They work mainly in the body by interacting with the different types of receptors in the central nervous system (CNS).
Here are some alkaloid-producing plants you’re probably familiar with: coffee plant, coca leaf, kratom plant, tobacco plant, poppy flower, tridax daisy.
Alkaloids & Plant Life
In plants, alkaloids can be found in many different areas, such as the bark, leaf, root, seed, fruit, and flower, and they are known as secondary metabolites, meaning they aren’t required for the growth and development of the plant. Primary metabolites, which are required for these purposes, are carbohydrates, fats, and proteins. Other secondary metabolites include tannins, resin, gums, and latex.
It’s theorized that alkaloids developed as a means to create advantages for a plant organism by either increasing its survivability or its fecundity. However, there are many cases in which the function of the alkaloids vis-à-vis the plant they can be found in is not clear. In most plants, alkaloids serve a protective function by defending the plant against natural predators. These types of alkaloids are toxic and protect the plant by making it inedible. Many alkaloids are also known to regulate plant growth.
There are some interesting cases of animals evolving to adapt to the consumption of certain alkaloids and even using them to attain competitive advantages. For instance, the ornate moth (Utetheisa ornatrix) will use the pyrrolizidine alkaloid to render itself unpalatable to a wide array of its natural predators.
Related: Can Fertilizers Alter Kratom Alkaloids?

Kratom Alkaloids
At the moment, scientists have successfully identified 54 different alkaloids in the leaves of the kratom plant [1]. This is quite a high number when compared to other popular alkaloid-producing plants like coffee and tobacco.
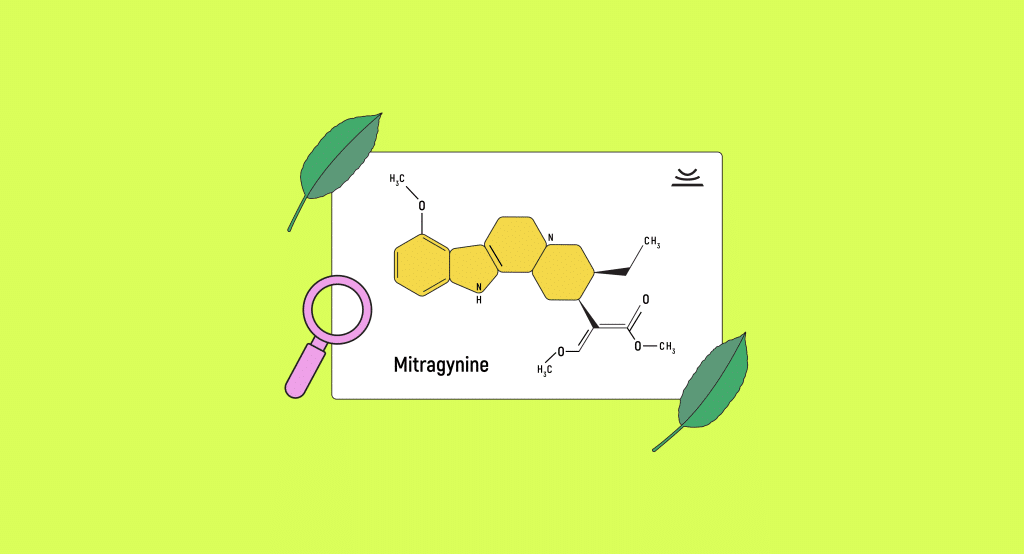
Mitragynine
The most abundant alkaloid in kratom and the one most responsible for its effects is mitragynine [2].
Mitragynine and its complementary alkaloid 7-hydroxymitragynine have been shown to modulate opioid receptors. They’re partial agonists at mu-opioid receptors and competitive antagonists at kappa and delta-opioid receptors [3]. One of their most important qualities is how they don’t activate the β-arrestin pathway, which is responsible for some of the most dangerous side effects of traditional opioids like respiratory depression and sedation [4].
Seeing as how mitragynine is the primary alkaloid responsible for kratom’s effects, we can also learn much about their potential uses by studying the benefits of kratom.
In low doses, kratom acts as a stimulant, providing boosts of energy and focus to the user. It also has euphoric properties. As the dose of kratom increases, the effects begin to shift towards wholly distinct benefits.
Mid-to-high doses of kratom will provide analgesia, as well as anxiolytic (anti-anxiety) relief. But that’s not all; kratom has also been used effectively as sleeping support and a weight loss supplement. It can even ameliorate the effects of opioid withdrawals!
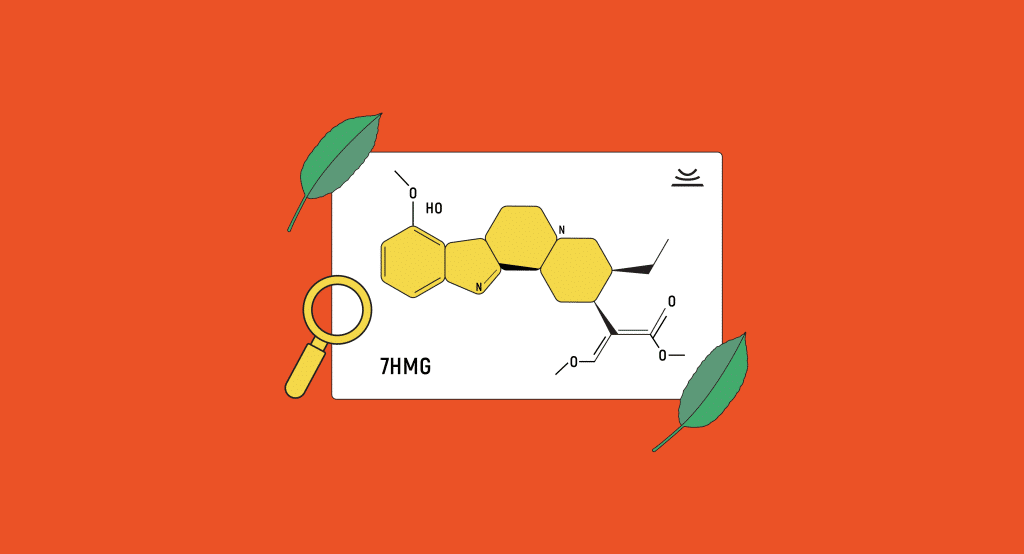
7-hydroxymitragynine
7-hydroxymitragynine, although not as abundant, is also quite important in producing kratom’s properties. It is considered to be an oxidized derivative and active metabolite of mitragynine.
It only accounts for about 2% of the alkaloid content, but studies have shown that 7-hydroxymitragynine is an incredibly potent compound, and so it has an outsized effect on kratom’s properties. 7-hydroxymitragynine reportedly has 13 times the strength of morphine and 46 times the strength of mitragynine.
Studies have shown that 7-hydroxymitragynine is a key compound in meditating kratom’s analgesic effects via the mu-opioid receptor [5].
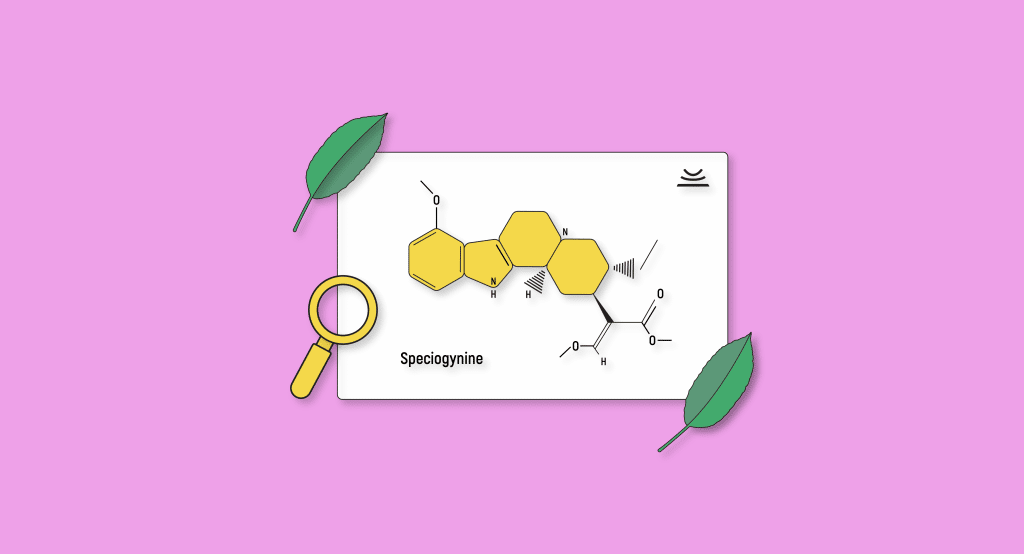
Speciogynine
Speciogynine is a diastereoisomer of mitragynine. These are compounds that have the same basic molecular formula and sequence of bonded elements but are not mirror images of each other. These types of isomers occur when two or more stereoisomers have different configurations.
As mentioned earlier, kratom research is limited, and there is simply not a lot of information available about the pharmacological effects of speciogynine. What few studies are available tend to focus more on identifying the alkaloids in kratom and determining their precise chemical structure.
Speciogynine compromises around 6-8% of the alkaloids found in kratom — although it should be noted that large discrepancies in alkaloid presence have been identified in numerous studies. These differences in alkaloid content can vary due to a lot of factors, like the type of kratom strain, the maturation of the kratom leaves at the time of harvest, the soil composition, etc.
Given that speciogynine shares the same basic formula as mitragynine, it’s probably not a stretch to assume that it holds similar pharmacological properties. As a rule of thumb, strains with high levels of mitragynine will probably be high in speciogynine.
We know that speciogynine is a weak opioid receptor agonist, so it likely has a role to play in giving the kratom plant its analgesic effects. Speciogynine is assuredly less potent than mitragynine and 7-hydroxy-mitragynine, but we can’t rule out its importance, as it might have an important synergic role [4]. Kratom is a dose-dependent drug, so speciogynine will probably be a mild stimulant as well.
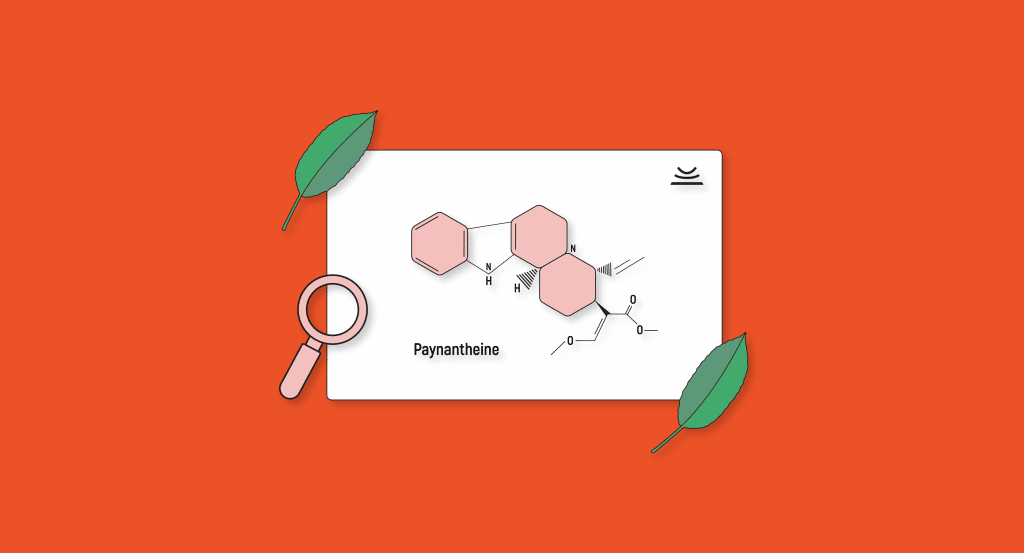
Paynantheine
Paynantheine is another derivative of mitragynine and a significant alkaloid in kratom’s alkaline profile. In terms of its effects, paynantheine has been found to have similar properties as that of 7-hydroxy mitragynine, although it’s not as significant. Unfortunately, this was assessed through indirect means as there are no direct studies attempting to ascertain paynantheine’s pharmacological effects.
Paynantheine has been found to have two diastereoisomers in the kratom plant, which likely have similar effects: isopaynantheine and epiallo-isopaynantheine [6].
Kratom naturally contains paynantheine, which forms as the result of metabolic processes involved in the creation of other alkaloids. Additionally, a study found very small concentrations of paynantheine in the kratom plant, enough to be considered one of the minor alkaloids in kratom in terms of content.
Research has determined that paynantheine is a competitive antagonist at the mu-opioid receptor and that it has a certain affinity with 7-hydroxy mitragynine. The connection between these two alkaloids suggests that strains with high 7-hydroxymitragynine are likely to contain considerable levels of paynantheine as well. Since we know that red-veined kratom strains have high levels of 7-hydroxy mitragynine, you should probably turn to this type of strain if you’re interested in paynantheine.
Red-veined kratom strains excel at producing analgesic benefits. Many patients suffering from chronic or acute pain often use them and even drop their pharmacological pain killers for them. Red vein kratom strains also have anxiolytic and sleep support properties. Studies have confirmed that paynantheine is a mu-opioid receptor antagonist, which gives further evidence for their linkage to red-veined strains.
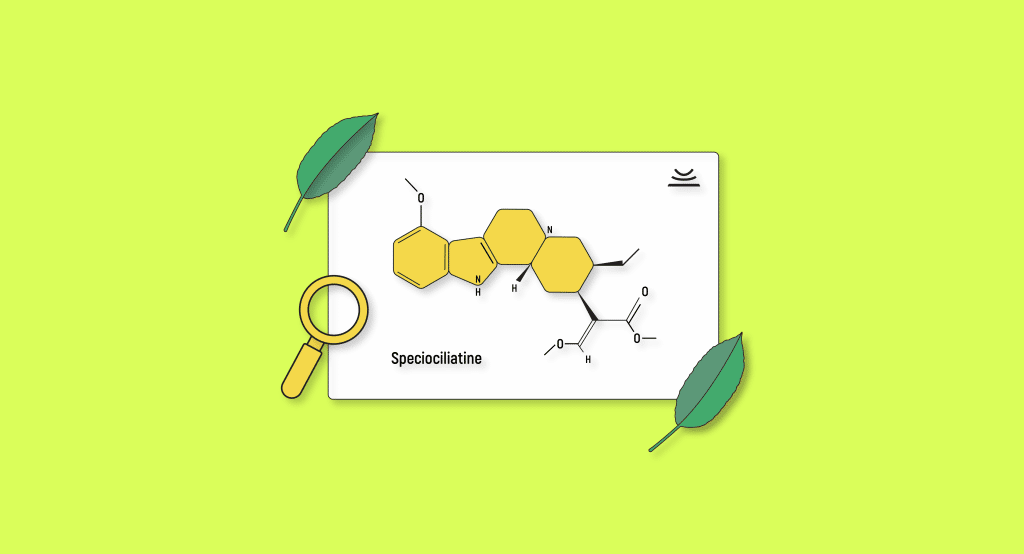
Speciociliatine
Despite being the second most numerous alkaloid in several kratom strains, speciociliatine is considered to be a secondary alkaloid in the kratom plant. It typically accounts for 8.6 percent of alkaloid content. Additionally, recent studies suggest that speciociliatine could be useful as a biomarker for kratom, facilitating the confirmation of kratom presence in a biological sample.
Similar to speciogynine, speciociliatine is also a diastereoisomer of mitragynine. What little research there is also tells us that speciociliatine acts mainly as an analgesic, which means that in red vein kratom strains, it is likely found in larger quantities.
Suppose you’re specifically looking to ingest more speciociliatine. In that case, we recommend the kratom strains with the most potent analgesic qualities: Red Maeng Da kratom, Red Borneo kratom, and Red Kali kratom.
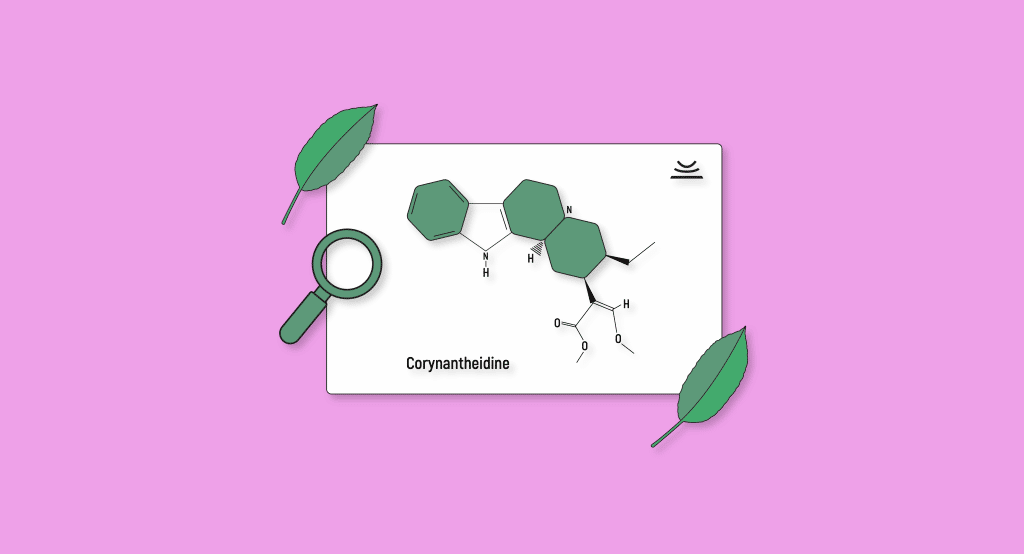
Corynantheidine
Corynantheidine is one of the least studied alkaloids found in kratom that is still thought to be significant. It only shows up in exceedingly small concentrations, making isolation and research a hard task. In practice, this means that there are virtually no studies looking at the pharmacological effects of corynantheidine.
Like the other secondary kratom alkaloids, corynantheidine is an opioid receptor antagonist and likely contributes to kratom’s analgesic and sleep support properties.
We also know that corynantheidine is able to interact with the 5HT2A receptors. This suggests that corynantheidine also plays a role in kratom’s euphoric properties. These receptors are also targeted by drugs such as MDMA, LSD, psilocybin (magic mushrooms), and other psychedelics with potent euphoric effects.
Other studies have revealed how corynantheidine can behave as an α1-adrenergic and α2-adrenergic receptor antagonists. These receptors have been shown to make us feel more alert and energized when activated. Conversely, if inhibited, they can also make us more lethargic and sedated. We could thus speculatively assume that corynantheidine may interact with these receptors to contribute to kratom’s stimulant or relaxant effects.
It seems that the secondary metabolites which contribute to kratom’s “mind” based effects (i.e., increased mental focus and euphoric properties) are somewhat harder to identify. Still, corynantheidine appears to be one of them. Another possibility is the dose-dependent quality of kratom, which isn’t well understood as of today.

Minor Kratom Alkaloids
The alkaloids above are considered to be secondary alkaloids, but at least something is known about them. However, there is a whole other swathe of alkaloids of which we basically know little to nothing about.
Alkaloids in this group, such as mitraciliatine, mitraphylline, and isomitraphylline, typically account for less than 1 percent of total alkaloid content collectively.
When we look at all of the 54 alkaloids that have been identified, we can group them into two overarching categories: indole alkaloids of oxindole alkaloids.
Indole alkaloids contain the structural formula of indole, and they’re considered to be one of the largest alkaloid groups with over 4100 known compounds. Much more is known about this group as many of them contain significant pharmacological and psychotropic potential and have been used by humans for thousands of years.
Much less is known about oxindole alkaloids, although studies have confirmed their chemical structure and have found they contribute considerably to sedation. Other studies have also found they exhibit neuroprotective and anti-inflammatory benefits [7].
Comprehensive List of Kratom Alkaloids
Lastly, here is a comprehensive list of all the alkaloids that have been identified in the kratom plant
Indole alkaloids:
- Mitragynine
- Mitragynine-N(4)-oxide
- 7-hydroxymitragynine
- Speciogynine
- Paynantheine
- Mitralactonal
- Speciociliatine-N(4)-oxide
- Isopaynantheine-N(4)-oxide
- 3,4,5,6,N-tetradehydromitragynine
- 7-hydroxymitraciliatine
- Mitragynine
- Mitraciliatine
- Corynantheidine
- Isopaynantheine
- 9-methoxycorynantheidinalinic acid
- Mitralactonine
- Ajmalicine
- Epiallo-Isopaynantheine-N(4)-oxide
- Mitrasulgynine
- 3-Dehydromitragynine
- 7-hydroxyspeciociliatine
- Corynantheidaline
- Speciociliatine
- Isocorynantheidine
- Epiallo-isopaynantheine
- Corynantheidinalinic acid
- Mytragynalinic acid
- 9-Metoxymitralactonine
Oxindole alkaloids:
- Mitragynine oxindole A
- Rotundifoleine
- Corynoxine A
- Isorhynchophylinne
- Mitrafoline
- Rhynchociline
- Mitraphylline
- Isospecionoxeine
- Isocoynoxeine
- Isorotundifoleine
- Corynoxine B
- Rhynchophylline
- Isomitrafoline
- Isospeciofoline
- Mitragynine oxindole B
- Speciophylline
- Specionoxeine
- Corynoxeine
- Isospeciofoleine
- 3-epicorynoxine B
- 3-epirhynchophylline
- Rotundifoline
- Isorotundifoline
- Speciofoline
- Ciliaphyline
- Isomitraphylline

Other Examples of Plant-Derived Alkaloids
Here are some other examples of major plant-derived alkaloids and their properties.
1. Caffeine
Surely we’ve all heard of this alkaloid.
Caffeine is a purine alkaloid derived from the coffee plant. It is used by humans as a central nervous stimulant of the methylxanthine class.
Caffeine has several mechanisms of action. For example, it increases energy metabolism throughout the brain but decreases at the same time cerebral blood flow, inducing relative brain hypoperfusion. Caffeine also activates noradrenaline neurons and seems to affect the local release of dopamine [8]. Lastly, caffeine also promotes wakefulness by blocking adenosine receptors and thus preventing the drowsiness induced by this compound.
Evidence also points to caffeine’s usefulness as a cognitive enhancer, increasing alertness and attention [9].
Also see: All You Need to Know About Caffeine Pills
2. Capsaicin
Capsaicin is an alkaloid derived from chili peppers (plants belonging to the genus Capsicum) and is the compound that gives them their spicy properties.
As the natural compound responsible for the spiciness of chili peppers, capsaicin obviously has a wide breadth of applications in food, but it also has medicinal properties.
Capsaicin is used in certain ointments and dermal patches for its analgesic properties [10]. It has also been approved by the FDA for medications that reduce the symptoms of peripheral neuropathy, such as post-herpetic neuralgia caused by shingles [11].
3. Cocaine
Derived from the coca leaf, cocaine is a tropane alkaloid with medicinal uses, although it is most commonly used as a recreational drug. It is a powerful central nervous system stimulant and has a known potential for abuse. However, it should be noted that recreational cocaine is created with a vast number of dangerous adulterants.
Medicinally, cocaine is mainly used as a numbing agent in surgical procedures involving the mouth or nose [12]. It also serves the dual role of vasoconstriction to reduce bleeding.

4. Ephedrine
Ephedrine is an alkaloid derived from several plants in the genus Ephedra. It works primarily by increasing the activity of norepinephrine (noradrenaline) on adrenergic receptors [13].
Ephedrine has many different medical uses. Mainly, it is used to prevent low blood pressure during anesthesia, but it has also been used for asthma, narcolepsy, and obesity [13].
5. Ergotamine
Ergotamine is part of the ergot family of alkaloids. It’s produced by the fungus genus Claviceps found in cereal kernels and grass seeds [14].
Ergotamine is used by doctors in the treatment of acute migraine attacks, as well as clustered headaches [15]. It’s available as a suppository, sublingual tablet, and regular tablet.
6. Mescaline
Mescaline is a psychedelic alkaloid of the substituted phenethylamine class. It can be found in different types of cacti and certain beans. Mescaline is commonly known for its hallucinogenic effects.
There is a medicinal interest in mescaline as it has been shown to have the potential to treat depression [16]. Some researchers also believe it can treat alcoholism.
Unfortunately, scientific research on mescaline is rare because of its status as a controlled substance.

7. Morphine
Morphine is one of several alkaloids derived from the poppy plant. It is a very effective compound in relieving moderate to severe pain. It’s also used for several preoperatory purposes like relieving anxiety and sedation [17].
However, it should be noted that morphine has a known potential for misuse. In the United States, it is classified as a Schedule II drug in the Controlled Substances Act.
8. Muscimol
Muscimol, an alkaloid, is derived from the Amanita muscaria mushroom and is one of the primary compounds responsible for its psychoactive effects. Muscimol is a powerful selective orthosteric agonist for the GABAA receptors and possesses sedative-hypnotic and depressant properties [18].
Trials have been conducted to study muscimol’s effectiveness in the treatment of epilepsy and Parkinsons’s disease. However, it has yet to receive FDA approval.
9. Quinine
The quinine alkaloid is found in the bark of the cinchona tree, commonly located in South America.
Medically, it is used to treat malaria and babesiosis [19]. It has been used for this purpose for hundreds of years but it is still not clear how it achieves this effect within the body.
10. Scopolamine
Scopolamine is a naturally occurring alkaloid produced by certain plants in the nightshade family. This compound is used mainly for the treatment of motion sickness, postoperative nausea, and vomiting [20].
11. Theobromine
Theobromine is the primary alkaloid found in the cacao plant and can usually be found in chocolate. It can also be found in other plants.
Theobromine has a similar structure to caffeine and has very similar, although somewhat lessened, effects. The main mechanisms of action are inhibition of phosphodiesterases and blockade of adenosine receptors [21].

Synthetic Alkaloids
Many alkaloids, especially those with important medical applications, are copied by scientists in a laboratory setting. These are called biomimic alkaloids. However, certain synthetic-only substances have similar structures to plant-based alkaloids. These are called synthetic alkaloids.
Synthetic alkaloids usually have similar functions and uses to their organic parent compounds but with slight variations that make them more suited to medical use.
Examples of Synthetic Alkaloids
These are some of the synthetic alkaloids produced by scientists:
- Procaine
- Buprenorphine
- Etorphine
- Methorphan
- Hydromorphone
The State of Kratom Research in The United States
Unfortunately, due to widespread governmental opposition in several countries, kratom is currently in the midst of various regulatory and legal difficulties. In the US, for example, kratom remains an unregulated substance, which hinders the ability to conduct scientific research on it for several reasons, one of the primary ones being the hesitancy of organizations to award scientific grants for kratom research.
With the possibility of kratom becoming a controlled substance, scientific institutions see little incentive to use their grant money to research it.
In practice, this means there’s not a lot of legitimate peer-reviewed research on kratom currently available. However, it’s not all bad news. The explosion in kratom’s popularity will attract more attention from the scientific community, and when you factor in the current opioid epidemic going on in the US, there’s a lot more incentive to start seriously looking at kratom and its benefits.
Related: Making Sense of Kratom Research.

Summary: Alkaloids in Kratom
Out of all these alkaloids, only 13 of them are commercially available. Unsurprisingly, these are the alkaloids that we know most about. However, the vast majority of research has been focused on mitragynine and 7-hydroxymitragynine.
The incredible variety of kratom’s alkaloid profile is most likely a huge contributor to its many benefits and properties. It also likely has something to do with kratom being a dose-dependent drug.
As kratom research matures, we’ll likely start to learn more about kratom’s secondary and minor alkaloids. Several scientists have posited that even though many alkaloids might have a minor role, they might still be quite important in the end result as they could have a significant synergic role.
It’s important to know that alkaloids are an incredibly vast classification of compounds. They are so naturally diverse that, in practice, there are still a lot of gray areas as to how they interact with each other in a complex biological system such as kratom.
- Flores-Bocanegra, L., Raja, H. A., Graf, T. N., Augustinović, M., Wallace, E. D., Hematian, S., … & Oberlies, N. H. (2020). The chemistry of kratom [Mitragyna speciosa]: Updated characterization data and methods to elucidate indole and oxindole alkaloids. Journal of natural products, 83(7), 2165-2177.
- Ya, K., Tangamornsuksan, W., Scholfield, C. N., Methaneethorn, J., & Lohitnavy, M. (2019). Pharmacokinetics of mitragynine, a major analgesic alkaloid in kratom (Mitragyna speciosa): A systematic review. Asian journal of psychiatry, 43, 73-82.
- Kruegel, A. C., & Grundmann, O. (2018). The medicinal chemistry and neuropharmacology of kratom: a preliminary discussion of a promising medicinal plant and analysis of its potential for abuse. Neuropharmacology, 134, 108-120.
- Ya, K., Tangamornsuksan, W., Scholfield, C. N., Methaneethorn, J., & Lohitnavy, M. (2019). Pharmacokinetics of mitragynine, a major analgesic alkaloid in kratom (Mitragyna speciosa): A systematic review. Asian journal of psychiatry, 43, 73-82.
- Kruegel, A. C., Uprety, R., Grinnell, S. G., Langreck, C., Pekarskaya, E. A., Le Rouzic, V., … & Sames, D. (2019). 7-Hydroxymitragynine is an active metabolite of mitragynine and a key mediator of its analgesic effects. ACS central science, 5(6), 992-1001.
- Kerrigan, S., & Basiliere, S. (2022). Kratom: A systematic review of toxicological issues. Wiley Interdisciplinary Reviews: Forensic Science, 4(1), e1420.
- Kushida, H., Matsumoto, T., & Ikarashi, Y. (2021). Properties, Pharmacology, and Pharmacokinetics of Active Indole and Oxindole Alkaloids in Uncaria Hook. Frontiers in Pharmacology, 12, 1769.
- Nehlig, A., Daval, J. L., & Debry, G. (1992). Caffeine and the central nervous system: mechanisms of action, biochemical, metabolic and psychostimulant effects. Brain Research Reviews, 17(2), 139-170.
- Wood, S., Sage, J. R., Shuman, T., & Anagnostaras, S. G. (2014). Psychostimulants and cognition: a continuum of behavioral and cognitive activation. Pharmacological Reviews, 66(1), 193-221.
- Fattori, V., Hohmann, M. S., Rossaneis, A. C., Pinho-Ribeiro, F. A., & Verri, W. A. (2016). Capsaicin: current understanding of its mechanisms and therapy of pain and other pre-clinical and clinical uses. Molecules, 21(7), 844.
- Fattori, V., Hohmann, M. S., Rossaneis, A. C., Pinho-Ribeiro, F. A., & Verri, W. A. (2016). Capsaicin: current understanding of its mechanisms and therapy of pain and other pre-clinical and clinical uses. Molecules, 21(7), 844.
- Dwyer, C., Sowerby, L., & Rotenberg, B. W. (2016). Is cocaine a safe topical agent for use during endoscopic sinus surgery?. The Laryngoscope, 126(8), 1721-1723.
- Abourashed, E. A., El‐Alfy, A. T., Khan, I. A., & Walker, L. (2003). Ephedra in perspective–a current review. Phytotherapy Research, 17(7), 703-712.
- Tfelt-Hansen, P., Saxena, P. R., Dahlöf, C., Pascual, J., Lainez, M., Henry, P., … & Goadsby, P. J. (2000). Ergotamine in the acute treatment of migraine: a review and European consensus. Brain, 123(1), 9-18.
- Zajdel, P., Bednarski, M., Sapa, J., & Nowak, G. (2015). Ergotamine and nicergoline—facts and myths. Pharmacological Reports, 67(2), 360-363.
- Agin-Liebes, G., Haas, T. F., Lancelotta, R., Uthaug, M. V., Ramaekers, J. G., & Davis, A. K. (2021). Naturalistic use of mescaline is associated with self-reported psychiatric improvements and enduring positive life changes. ACS Pharmacology & Translational Science, 4(2), 543-552.
- Wiffen, P. J., Wee, B., & Moore, R. A. (2016). Oral morphine for cancer pain. Cochrane Database of Systematic Reviews, (4).
- Johnston, G. A. (2014). Muscimol as an ionotropic GABA receptor agonist. Neurochemical Research, 39(10), 1942-1947.
- Achan, J., Talisuna, A. O., Erhart, A., Yeka, A., Tibenderana, J. K., Baliraine, F. N., … & D’Alessandro, U. (2011). Quinine, an old anti-malarial drug in a modern world: role in the treatment of malaria. Malaria Journal, 10(1), 1-12.
- Riad, M., & Hithe, C. C. (2021). Scopolamine. In StatPearls [Internet]. StatPearls Publishing.
- Martínez-Pinilla, E., Oñatibia-Astibia, A., & Franco, R. (2015). The relevance of theobromine for the beneficial effects of cocoa consumption. Frontiers in pharmacology, 30.






