Does Kratom Interact With Ondansetron (Zofran)?
Yes, kratom can interact with ondansetron when taken simultaneously. Ondansetron does not elicit any psychotropic effects and, as such, will not interfere with kratom’s properties.
However, these compounds do have a metabolic interaction.
Ondansetron is metabolized mainly by CYP3A4 with some involvement of CYP2D6 and CYP1A2 [1]. This is significant as kratom is a potent inhibitor of CYP2D6 and a moderate inhibitor of CYP3A4 [2].
The inhibition of ondansetron’s metabolization will increase the likelihood of adverse effects.
Is it Safe to Take Kratom With Ondansetron (Zofran)?
No. The metabolic interaction between kratom and antiemetics like ondansetron carries a mild to moderate risk.
Regarding metabolic inhibition, if you only plan to take ondansetron once, concomitant use with kratom will likely not result in significant risk. If you must use ondansetron daily, however, this will increase the risk factor considerably.
To stay safe, it’s best to avoid this combination or talk to your doctor first. Since ondansetron is used mainly when the patient is undergoing severe nausea and other negative symptoms, abstaining from kratom should not be difficult.

What is Ondansetron?
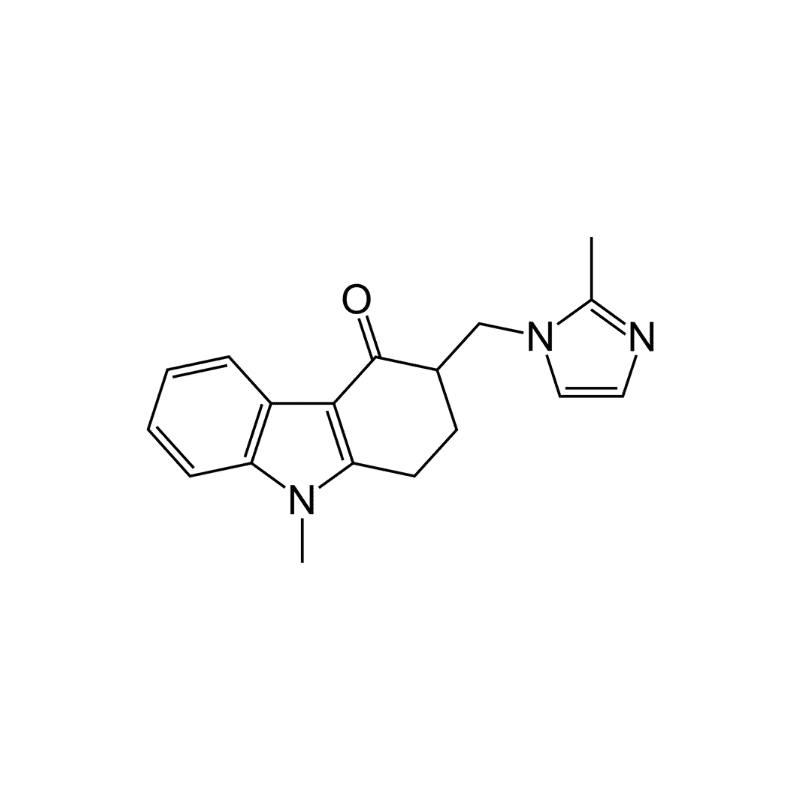
Ondansetron is a prescription-only antiemetic medication that is sold under the brand name Zofran.
There are several types of antiemetics.
Ondansetron is a serotonin 5-HT3 receptor antagonist that blocks serotonin receptors in the central nervous system and the gastrointestinal tract. This inhibits the action of serotonin — a natural substance that has a role in causing nausea and vomiting. It is available by tables, film, oral solution, and injection into a muscle or vein [3].
Ondansetron Specs
| Drug Name | Ondansetron |
| Trade Name | Zofran |
| Classification | Antiemetic |
| CYP Metabolism | CYP3A4, CYP2D6 and CYP1A2 |
| Interaction With Kratom | Metabolic inhibition |
| Risk of Interaction | Moderate |
What Is Ondansetron Used for?
The FDA has approved ondansetron for the following indications:
- Nausea and vomiting associated with initial and repeat courses of emetogenic cancer chemotherapy (including cisplatin)
- Postoperative nausea or vomiting
- Nausea and vomiting associated with radiotherapy in patients receiving either total body radiation, a single high-dose fraction to the abdomen, or daily fractions to the abdomen
Ondansetron also has several off-label uses (not approved by the FDA):
- Treatment of morning sickness and hyperemesis gravidarum during pregnancy [4]
- Cyclic vomiting syndrome
- Reduction of vomiting as associated with gastroenteritis [5]
Generic & Brand Name Versions
Ondansetron is available under the following brand names:
- Zofran
- Zuplenz
What’s the Dose of Ondansetron?
The recommended dose of ondansetron varies based on the ailment and the user’s age. The FDA recommends the following:
Adult doses:
- Highly emetogenic chemotherapy: A single 24 mg dose administered 30 minutes before the start of single-day highly emetogenic chemotherapy, including cisplatin greater than or equal to 50 mg/m2
- Moderately emetogenic chemotherapy: 8 mg administered 30 minutes before the start of chemotherapy, with a subsequent 8 mg dose 8 hours after the first dose
- Radiotherapy: usually 8 mg
- Postoperative: 16 mg administered 1 hour before induction of anesthesia.
Pediatric doses:
- Moderately emetogenic chemotherapy:
- (12-17): 8 mg administered 30 minutes before the start of chemotherapy, with a subsequent 8-mg dose 4 and 8 hours after the first dose.
- (4-11): 4 mg administered 30 minutes before the start of chemotherapy, with a subsequent 4 mg dose 4 and 8 hours after the first dose.
There is little clinical data on the use of ondansetron on people under 4 years of age. No recommended doses can be given in these instances.
Those with severe hepatic impairment should not exceed a daily dose of 8 mg.
What Are the Side Effects of Ondansetron?
The most common adverse effects of ondansetron use are [3, 6]:
- Abdominal pain
- Constipation
- Diarrhea
- Dry mouth
- Headache
- Malaise/fatigue
Serious side effects include:
- QT prolongation
- Serotonin syndrome
- Severe allergic reaction
There are also contraindications to taking ondansetron:
- Concomitant use of apomorphine
- Patients with a hypersensitivity to ondansetron or its components
Using ondansetron during pregnancy carries a low risk of harm to the baby and increases the risk of heart problems [7].

What is Kratom?
Kratom is an all-natural plant compound derived from the leaves of the Mitragyna speciosa, a tropical evergreen tree native to Southeast Asia.
Local indigenous peoples have used kratom for many centuries to aid them in their day-to-day tasks and as medicine. Now, the western hemisphere is finally noticing the plant’s many benefits.
Kratom’s effects are due to the many plant-based alkaloids it contains. These compounds interact with the opioid receptors in the body’s central nervous system to produce various effects.
Nevertheless, despite its many favorable properties, kratom can be addictive and has contributed to overdose deaths in the United States. However, this is rare and almost always results from interactions with other drugs.

What is Kratom Used for?
The effects of kratom depend on the amount ingested.
A lower dose promotes nootropic, mind-based effects like increased mental energy and mood-enhancing properties. Higher doses lead to more analgesic and sedative-like effects — ideal for chronic pain and relaxation.
Essentially, kratom’s properties may help to alleviate numerous ailments, but this plant can also serve as a nootropic supplement.
In terms of medication, kratom can help relieve chronic pain. It also has anxiolytic qualities and can help ease the symptoms of depression and anxiety.
Additionally, kratom has shown promise as a weight-loss supplement and works well while treating different withdrawal symptoms.
Only time will tell what other uses for kratom there are.
What’s the Dose of Kratom?
It’s crucial to get your kratom dosage right.
As we’ve just seen, the effects of the kratom plant are highly dependent on dosage. If you want to make the best of your kratom experience, choose the proper dosage for the job.
- Low dose (1 – 5 g): Produces nootropic and stimulant effects such as increased mental functioning and euphoria.
- Medium dose (5 – 10 g): Analgesic and anxiolytic properties become more evident in this range.
- High dose (10 – 15 g): heavy doses of kratom provide substantial pharmacological benefits and potent sedative-like effects.
Remember that everyone’s bodies are different, and formulaic dosage recommendations are generally best avoided.
Also see: How Much Kratom is Too Much?

What Are the Side Effects of Kratom?
Kratom and opioid medications have roughly the same side effects. However, kratom’s adverse effects are usually much less severe and can be avoided by starting with a low dose and slowly adjusting it.
Kratom’s common side effects are:
- Constipation
- Depression
- Dizziness
- Headaches
- Insomnia
- Itchiness
- Loss of muscle coordination
- Nausea
More severe side effects include:
- Liver damage
- Low blood pressure
- Low libido
- Poor appetite
- Seizures
- Tremors
Additionally, kratom consumption can cause an addiction, though its overdose potential is relatively low. Overall, kratom is safe and helpful for most people.
What Are the Different Types of Kratom?
There are four different kratom strains: this is excellent news for you if you’re the type who likes to personalize their experience!
These strains have the same basic properties, but each emphasizes specific effects.
Familiarize yourself with the different strains if you’re looking to take kratom for a specific purpose — it will help you get the most bang for your buck.

White Vein Kratom
White-veined kratom strains fall under the mind-based spectrum of kratom benefits. They stimulate mental focus, euphoria, and generalized states of increased mental activity.
White strains are likely your best option if you’re looking to try kratom for its stimulant-like properties.

Red Vein Kratom
Red vein kratom strains are ideal for anyone experiencing chronic pain but who doesn’t want pharmacological painkillers.
Red vein kratom also has anxiolytic benefits, and it’s popularly used as sleep support since it promotes calm and relaxation.

Green Vein Kratom
Green vein kratom sits right between white and red in the kratom spectrum.
This means that green-veined strains have a balanced alkaloid profile with analgesic and anxiolytic properties and mind-based effects.
However, this means that you won’t experience the targeted nature of a white or red kratom strain.

Yellow Vein Kratom
Yellow vein kratom is thought to be a mixture of red and white strains. For this reason, it is generally not considered one of the three main types of kratom.
Yellow vein users report that this strain is comparable to green kratom strains but has milder effects.

Key Takeaways: Is it Safe to Mix Kratom & Ondansetron (Zofran)?
Not necessarily. Due to their metabolic interaction, there’s an increased risk of adverse effects. It’s best to talk to your doctor before using kratom with ondansetron.
The main factor you must remember is that kratom consumption will lead to higher levels of ondansetron in the body due to its inhibitory metabolic effect. Luckily, ondansetron’s elimination half-life is relatively short (around 5 hours).
If you feel like you must continue using both compounds, then try to space them out and consult your doctor about what other alternatives are open to you.
- Bell, G. C., Caudle, K. E., Whirl-Carrillo, M., Gordon, R. J., Hikino, K., Prows, C. A., … & Schwab, M. (2017). Clinical Pharmacogenetics Implementation Consortium (CPIC) guideline for CYP2D6 genotype and use of ondansetron and tropisetron. Clinical pharmacology and therapeutics, 102(2), 213.
- Hanapi, N. A., Ismail, S., & Mansor, S. M. (2013). Inhibitory effect of mitragynine on human cytochrome P450 enzyme activities. Pharmacognosy research, 5(4), 241.
- Griddine, A., & Bush, J. S. (2022). Ondansetron. In StatPearls [Internet]. StatPearls Publishing.
- Siminerio, L. L., Bodnar, L. M., Venkataramanan, R., & Caritis, S. N. (2016). Ondansetron use in pregnancy. Obstetrics and gynecology, 127(5), 873.
- Freedman, S. B., Adler, M., Seshadri, R., & Powell, E. C. (2006). Oral ondansetron for gastroenteritis in a pediatric emergency department. New England Journal of Medicine, 354(16), 1698-1705.
- Bryson, J. C. (1992, December). Clinical safety of ondansetron. In Seminars in oncology (Vol. 19, No. 6 Suppl 15, pp. 26-32).
- Carstairs, S. D. (2016). Ondansetron use in pregnancy and birth defects: a systematic review. Obstetrics & Gynecology, 127(5), 878-883.